Medical Pharmacology Chapter 2 General Principles: Clinical Pharmacology
|
|
|
|
|
-
-
Audio Overview: Clinical Pharmacology (Extended)
Audio Overview: Clinical Pharmacology (Brief)
-
Introduction: Where Basic Science Meets the Patient
-
Clinical pharmacology is the discipline that bridges basic pharmacological science (the mechanisms, equations, and molecular interactions) and the practical art of prescribing for individual patients.
-
Clinical encounters involving a drug represents an application of these pharmacological principles:
-
Pharmacokinetics determines whether the drug reaches its target.
-
Pharmacodynamics determines what it does when it gets there, and
-
Clinical pharmacology principles determine whether the right drug has been chosen, at the right dose, for the right patient, at the right time.
-
-
Clinical Pharmacology considers principles that translate pharmacological knowledge into rational prescribing decisions and include:
-
Therapeutic decision-making framework
-
Concepts of rational prescribing, monitoring of drug therapy
-
Principles of drug use in special populations, and
-
Clinical consequences of pharmacological interactions.
-
These principles apply throughout clinical practice regardless of specialty,1,2
-
-
-
-
Therapeutic Decision-Making Framework
-
Before prescribing any drug, a structured clinical reasoning process should be applied.
-
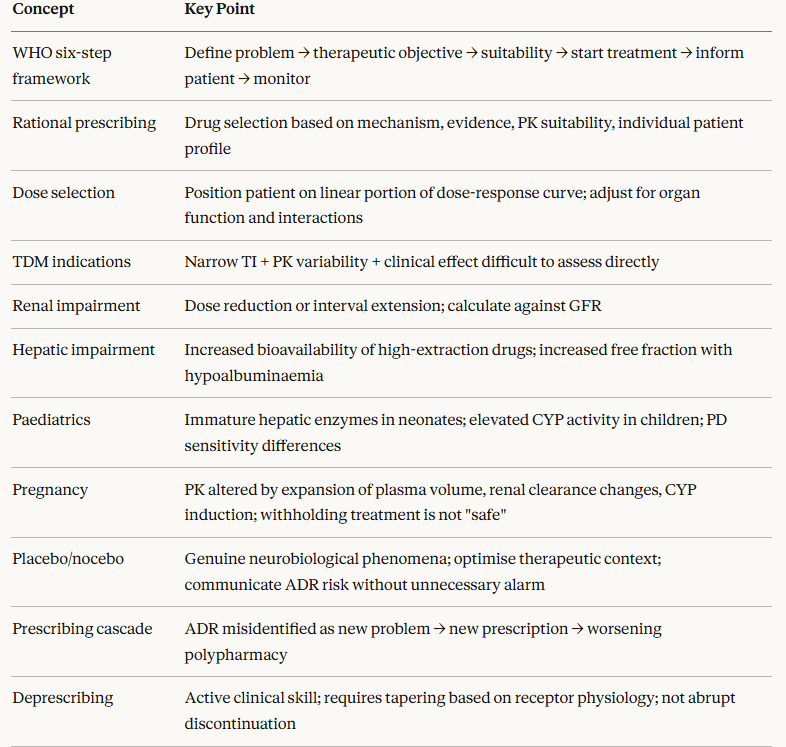
The WHO's Guide to Good Prescribing, widely adopted in medical curricula internationally, provides a six-step framework that makes the pharmacological basis of prescribing explicit.3
-
Step 1 Define the patient's problem
-
What is the diagnosis or clinical problem being treated?
-
Precision of the therapeutic decision follows from precision of the diagnosis.
-
Prescribing for imprecise problems (e.g., "dizziness," "fatigue") without a defined mechanism may lead to ineffective or harmful treatments.
-
-
-
Step 2 Specify the therapeutic objective
-
What do you want to achieve : cure, symptom control, prevention, or palliation?
-
Therapeutic objective determines the appropriate drug class.
-
An antibiotic for bacterial meningitis is prescribed to cure.
-
An antiemetic for chemotherapy-induced nausea is prescribed for symptom control.
-
Aspirin in a patient after myocardial infarction is prescribed for secondary prevention.
-
-
-
-
Step 3 Verify whether the chosen treatment is suitable for this patient
-
Does the drug's pharmacological profile match the patient's physiology?
-
The suitability assessment encompasses:
-
Contraindications
-
Drug interactions
-
Renal and hepatic function
-
Age
-
Pregnancy status, and
-
Pharmacogenomic considerations
-
-
-
-
 Select
the drug, dose, route, frequency, and
duration.
Select
the drug, dose, route, frequency, and
duration.
-
All four are pharmacologically determined decisions.
-
-
-
Step 5 Give information, instructions, and warnings
-
Patient education directly affects adherence and safety.
-
A patient who does not understand why they are taking a drug, what symptoms indicate toxicity, or how to take it correctly is a patient at elevated risk of treatment failure or adverse drug reactions.
-
-
-
Drug therapy is not a set-and-forget intervention.
-
Therapy requires active monitoring for therapeutic efficacy, adverse effects, and evolving patient circumstances that change the pharmacokinetic or pharmacodynamic context of the prescription.
-
-
-
-
Rational Prescribing: The Pharmacological Foundation
-
Rational prescribing is the selection and use of drugs based on evidence of efficacy, pharmacological appropriateness for the individual patient, and consideration of safety and cost relative to alternatives.
-
 This
approach to prescribing is the goal of clinical pharmacology as
a discipline and distinguishes pharmacologically grounded
therapeutic decisions from habit-based, marketing-influenced, or
defensive prescribing.1,2
This
approach to prescribing is the goal of clinical pharmacology as
a discipline and distinguishes pharmacologically grounded
therapeutic decisions from habit-based, marketing-influenced, or
defensive prescribing.1,2 -
The pharmacological elements of a rational prescribing decision include:
-
-
Does the drug's mechanism of action match the pathophysiology being treated?
-
An ACE inhibitor is rational in a patient with heart failure and hypertension because its mechanism (reducing angiotensin II-mediated vasoconstriction and aldosterone release) directly addresses two pathophysiological drivers.
-
Prescribing it in a patient with dihydropyridine calcium channel blocker-induced peripheral edema would be mechanistically inappropriate as the edema is vasodilatory, not cardiogenic.
-
-
-
-
Does the drug reach the target site in adequate concentrations given the patient's physiology?
-
A drug with poor CNS penetration will not treat a CNS infection regardless of its in vitro potency.
-
An orally administered drug will be ineffective in a patient who cannot absorb it due to gastrointestinal dysmotility or post-surgical anatomy.
-
-
-
-
-
Rational dose selection derives from the dose-response relationship and the individual patient's pharmacokinetic profile.
-
The dose should position the patient on the linear portion of the dose-response curve, above the threshold for efficacy but below the concentration where toxicity risk rises steeply.
-
Organ impairment, pharmacogenomic factors, and co-medications all shift this position.
-
-
-
-
-
Within a drug class that is appropriate for the indication, secondary pharmacological considerations including receptor subtype selectivity, drug interaction profile, duration of action, pharmacokinetic characteristics differentiate individual drugs.
-
These differences are pharmacologically meaningful.
-
-
-
-
-
Therapeutic drug monitoring (TDM) is the practice of measuring drug concentrations in blood and adjusting doses to maintain concentrations within a defined therapeutic range.
-
TDM is indicated when:
-
(1) There is a well-defined relationship between plasma drug concentration and therapeutic or toxic effect
-
(2) The therapeutic index is narrow (the margin between efficacious and toxic concentrations is small)
-
(3) Pharmacokinetic variability between patients is substantial
-
(4) Clinical assessment of drug effect is difficult or delayed
-
(4) Drug interactions or organ impairment significantly alter expected pharmacokinetics4
-
Therapeutic Drug Monitoring 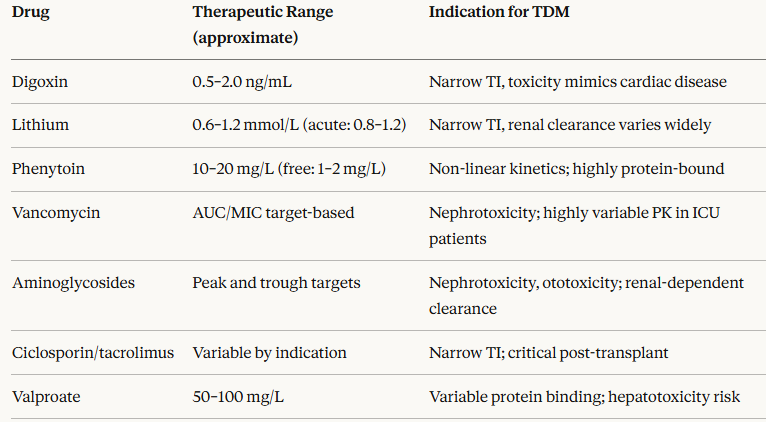
-
TDM results must always be interpreted in clinical context.
-
A "subtherapeutic" level in a patient with good disease control does not mandate a dose increase.
-
A "therapeutic" level in a patient with drug toxicity does not guarantee that the drug is not responsible.
-
The sample timing relative to the dose (trough vs peak vs AUC) is critical for correct interpretation.4
-
-
-
-
-
Pharmacological Basis of Drug Use in Special Populations
-
The principles of pharmacokinetics and pharmacodynamics apply universally, but their quantitative expression differs systematically in several patient populations, requiring deliberate modification of standard prescribing.1,2
-
-
 Drugs excreted predominantly unchanged by the kidney accumulate when GFR
falls.
Drugs excreted predominantly unchanged by the kidney accumulate when GFR
falls.
-
The required dose adjustment is proportional to the reduction in renal clearance.
-
Three strategies are employed: dose reduction (same interval, smaller dose), interval extension (same dose, longer interval), or both.
-
The choice depends on the drug's pharmacodynamic profile.
-
For antibiotics, where peak concentrations matter (aminoglycosides), interval extension maintains the peak; for drugs where continuous exposure is required (antiretrovirals), dose reduction is preferred.
-
Calculating the adjusted dose requires an estimate of residual GFR using the Cockcroft-Gault or CKD-EPI equation.
-
-
-
-
-
-
-
-
 Reduced
hepatic blood flow, impaired CYP enzyme activity, reduced albumin
synthesis, and portal-systemic shunting all alter drug disposition.
Reduced
hepatic blood flow, impaired CYP enzyme activity, reduced albumin
synthesis, and portal-systemic shunting all alter drug disposition.
-
For drugs with high hepatic extraction (e.g., morphine, lidocaine, propranolol), where oral bioavailability is normally very low due to first-pass metabolism, hepatic impairment dramatically increases oral bioavailability.
-
For drugs extensively bound to albumin (warfarin, phenytoin), hypoalbuminaemia increases the free fraction, elevating pharmacodynamic effect at apparently normal total plasma concentrations.
-
The Child-Pugh score classifies hepatic impairment and guides dose adjustment.
-
-
-
-
 Pharmacokinetics
in children are not simply scaled-down adult pharmacokinetics.
Pharmacokinetics
in children are not simply scaled-down adult pharmacokinetics.
-
Neonates have immature hepatic enzyme systems (CYP3A4, UGT) and reduced renal clearance relative to body surface area.
-
As children mature, hepatic CYP activity initially exceeds adult levels before declining toward adult norms, meaning that school-aged children may require higher weight-adjusted doses of some drugs (e.g., methylphenidate, anticonvulsants) than adults to achieve equivalent plasma concentrations.
-
Pharmacodynamic sensitivity also differs.
-
For example, neonates are more sensitive to opioid-induced respiratory depression because of immature blood-brain barrier function and CNS pharmacodynamic sensitivity.
-
-
-
-
-
-
Prescribing in pregnancy must balance the risk to the fetus against the risk of untreated maternal disease.
-
Pharmacokinetic changes of pregnancy include:
-
Expanded plasma volume (diluting drug concentrations)
-
Increased renal blood flow (accelerating clearance of renally excreted drugs)
-
Altered CYP expression (CYP3A4 and CYP2D6 induced; CYP1A2 reduced), and
-
Altered protein binding can reduce plasma drug concentrations below therapeutic levels at standard doses.
-
-
Active epilepsy is more dangerous to the fetus than most antiepileptic drugs
-
Severe depression and psychosis in pregnancy require treatment.
-
The principle is that withholding treatment is not necessarily "safe" but may be just substituting one risk for another.
-
-
-
-
-
Placebo Effects and the Clinical Context of Drug Action
-
No account of clinical pharmacology is complete without acknowledging that observed therapeutic responses to drugs are not purely pharmacological.
-
The placebo response representing improvement in outcomes attributable to expectation, therapeutic context, patient-clinician relationship, and neurobiological mechanisms triggered by treatment administration can be substantial and measurable.
-
In analgesic and antidepressant trials, placebo response rates of 30–50% are common.5
-
-
-
This is not a problem to be eliminated as it is a clinical phenomenon with a biological foundation.
-
Placebo analgesia has been shown to be mediated partly by endogenous opioid release (it is reversed by naloxone), possibly suggesting of neurobiological mechanism rather than merely biased reporting.
-
The clinical implication is that the pharmacological effect of a prescribed drug operates in a context of expectation, trust, and therapeutic environment and optimizing these factors is likely good clinical pharmacology.
-
-
The nocebo effect which is induction of adverse effects by negative expectation results in the inverse clinical implication.
-
That is, patients who are told to expect side effects are more likely to experience them.
-
Ccommunication of adverse drug effect risk should be accurate and complete, but not unnecessarily alarming which could itself cause harm.
-
-
Prescribing in an Era of Polypharmacy
-
Polypharmacy, the concurrent use of multiple drugs, typically defined as five or more, is now the norm rather than the exception in clinical practice, reflecting an aging population with multiple chronic conditions.
-
The pharmacological challenges of polypharmacy are not simply additive: drug interaction risk increases non-linearly with the number of co-prescribed drugs.2
-
The principles of rational prescribing require applying pharmacological reasoning explicitly in the polypharmacy context:
-
-
Each drug on a patient's medication list should be questioned periodically "is it still indicated"?
-
Is the original indication still present?
-
Is the evidence base for its use still supporting its continuation?
-
The START/STOPP criteria and Beers Criteria provide structured frameworks for identifying inappropriate prescribing in older patients.
-
-
-
-
-
A common pattern in polypharmacy is the prescribing cascade in which an Adverse Drug Response is misidentified as a new clinical problem and treated with a further prescription, rather than the causative drug being identified and withdrawn.
-
A β-blocker causes fatigue; fatigue is attributed to depression and an antidepressant is prescribed.
-
The SSRI causes bruxism and bruxism is attributed to temporomandibular joint dysfunction and an NSAID is prescribed.
-
 Recognizing
the prescribing cascade requires asking: "Could this new symptom be
caused by one of the patient's current medications?"
Recognizing
the prescribing cascade requires asking: "Could this new symptom be
caused by one of the patient's current medications?"
-
-
-
-
-
-
Principled, supervised withdrawal of drugs that are no longer indicated, have ceased to benefit, or whose risks now outweigh their benefits, is an active clinical pharmacology skill.
-
Abrupt discontinuation of long-term medications risks withdrawal syndromes (beta-blockers, benzodiazepines, glucocorticoids, opioids, SSRIs) which is an expectable pharmacodynamic consequence of receptor regulation.
-
Deprescribing requires planned tapering schedules based on receptor physiology.
-
-
-
-
-
 |
References
|
|