|
press  above to
begin the lecture above to
begin the lecture
and
install current free versions of Quicktime,
if needed, to support lecture series audio!
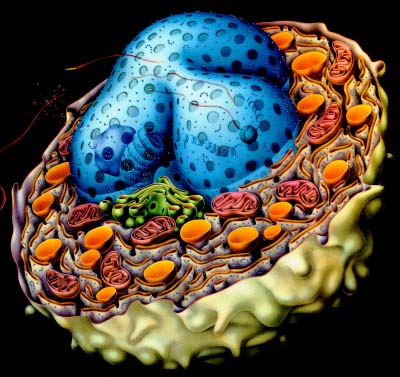
Figure Developed by Dr. Steve Downing, University of
Minnesota, illustrating the many membrane barriers even within a
single cell.
-
Absorption
-
Routes of
Administration
-
First-Pass Effect
-
Pulmonary
Effects
-
Pharmacokinetics
|
-
Drug
Metabolism
-
Introduction
-
Phase
I and Phase II Reaction Overview:
-
Phase
I characteristics
-
Phase
II characteristics
-
Conjugates
-
Principal
organs for biotransformation
-
Bioavailability
-
Microsomal
Mixed Function Oxidase System and Phase I Reactions
-
Phase II Reactions
-
Individual
Variation in Drug Responses
-
Genetic
Factors in Biotransformation
-
Effects
of Age on Drug Responses
-
Drug-Drug
Interactions
Pharmacokinetics
and some IV Anesthetics Agents
-
Barbiturates
-
Benzodiazepines
-
Ketamine
and Etomidate
-
Propofol
-
Opioids
|
-
II.
Lipid diffusion
-
Most important barrier for
drug permeation due to:
-
Lipid: aqueous drug
partition coefficients described the ease
with which a drug moves between aqueous
and lipid environments
-
Ionization state
of the drug is an important factor:
charged drugs diffuse-through lipid
environments with difficulty.
-
pH and the drug
pKa, important in determining the
ionization state, will influence
significantly transport (ratios
of lipid-to aqueous-soluble forms
for weak acids and bases
described by the Henderson-Hasselbalch equation.
-
uncharged
form: lipid-soluble
-
charged
form: aqueous-soluble, relatively
lipid-insoluble (does not
pass biological membranes
easily)
Henderson-Hasselbalch
equation
General Form: log (protonated)/(unprotonated) =
pKa - pH
- For Acids: pKa =
pH + log (concentration [HA]
unionized)/concentration [A-])
- note that if [A-] = [HA] then
pKa = pH + log (1) or (since log(1) = 0), pKa
= pH
|
- For Bases: pKa
= pH + log (concentration [BH+]
ionized)/concentration [B])
- note that if [B] = [BH+] then pKa
= pH + log (1) or (since log(1) = 0), pKa
= pH
|
-
The lower the pH relative to the
pKa the greater fraction of protonated drug is
found. Recall that the protonated form of an acid is
uncharged (neutral); however, protonated form of a base
will be charged.
-
As a result, a weak acid at acid
pH will be more
lipid-soluble because it is uncharged and uncharged
molecules move more readily through a lipid (nonpolar)
environment, like the some membrane, than charged
molecules
-
Similarly a weak base at alkaline
pH will be more
lipid-soluble because at alkaline pH a proton will
dissociate from molecule leaving it uncharged and again
free to move through lipid membrane structures

Many
drugs are weak acids or weak bases
-
A weak acid is
a neutral molecule that
dissociates into an anion
(negatively charged) and a proton
(a hydrogen ion) Example:
-
C8H7O2COOH
< > C8H7O2COO-
+ H+
-
Neutral aspirin
(C8H7O2COOH)
in equilibrium with
aspirin anion (C8H7O2COO-
) and a proton (H+)
-
weak acid:
protonated form --
neutral, more
lipid-soluble
-
weak base: a neutral
molecule that can form a cation
(positively charged) by combining
with a proton. Example:
-
C12H11CIN3NH3+
< > C12H11CIN3NH2
+ H+
-
pyrimethamine
cation (C12H11CIN3NH3+)
in equilibrium
with neutral
pyrimethamine (C12H11CIN3NH2)
and a proton
(H+
)
-
weak base:
protonated form --
charged, less
lipid-soluble
|
| Weak
acids |
pKa |
weak bases |
pKa |
|
|
7.1 |
|
8.5 |
|
|
8.1 |
|
9.6 |
|
|
9.5 |
- chlordiazepoxide (Librium)
|
4.6 |
|
|
3.5 |
|
7.9 |
|
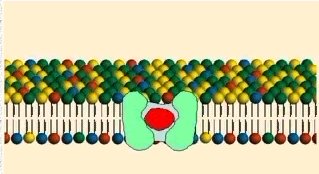
Figure Developed by Dr. Steve Downing,
University of Minnesota
Summary
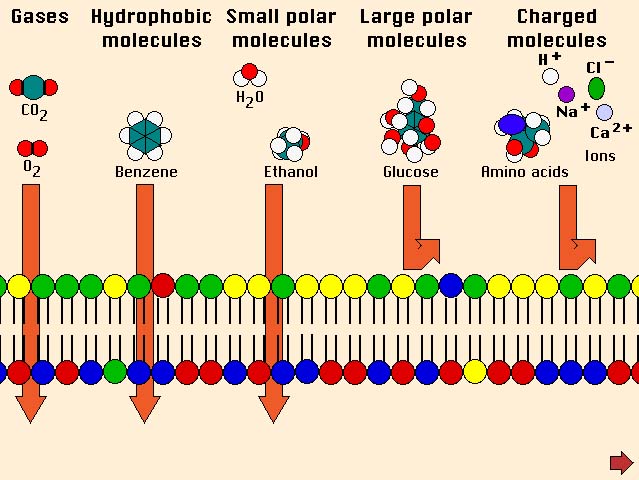
Figure Developed by Dr. Steve Downing, University of
Minnesota
|
Extent of Absorption
Ion Trapping
Ion Trapping: Anesthesia correlation:Placental
transfer of basic drugs
-
Placental transfer of basic
drugs from mother to fetus: local
anesthetics
-
Fetal pH is lower than
maternal pH
-
Lipid-soluble, nonionized
local anesthetic crosses the placenta
converted to poorly lipid-soluble ionized
drug
-
Gradient
is maintained for continual
transfer of local anesthetic from
maternal circulation to fetal
circulation
-
In fetal
distress, acidosis contributes to
local anesthetic accumulation
|

-
Weak bases-- amines
-
N + 1 carbon (R) and 2
hydrogens: primary amine (reversible protonation)
-
N + 2 carbons (R) and 1
hydrogen: secondary amine (reversible protonation)
-
N + 3 carbons (R):
tertiary amine (reversible protonation)
-
N + 4 carbons (R):
quaternary amine (permanently charged)
|
| Katzung, B. G. Basic
Principles-Introduction , in Basic and Clinical
Pharmacology, (Katzung, B. G., ed) Appleton-Lange, 1998,
pp 1-33 |
| Stoelting, R.K.,
"Pharmacokinetics and Pharmacodynamics of Injected
and Inhaled Drugs", in Pharmacology and Physiology
in Anesthetic Practice, Lippincott-Raven Publishers,
1999, 1-17. |
|