|
press  above to
begin the lecture above to
begin the lecture
and
install current free versions of Quicktime,
if needed, to support lecture series audio!
-
Absorption
-
Routes of
Administration
-
First-Pass Effect
-
Pulmonary
Effects
-
Pharmacokinetics
|
-
Drug
Metabolism
-
Introduction
-
Phase
I and Phase II Reaction Overview:
-
Phase
I characteristics
-
Phase
II characteristics
-
Conjugates
-
Principal
organs for biotransformation
-
Bioavailability
-
Microsomal
Mixed Function Oxidase System and Phase I Reactions
-
Phase II Reactions
-
Individual
Variation in Drug Responses
-
Genetic
Factors in Biotransformation
-
Effects
of Age on Drug Responses
-
Drug-Drug
Interactions
Pharmacokinetics
and some IV Anesthetics Agents
-
Barbiturates
-
Benzodiazepines
-
Ketamine
and Etomidate
-
Propofol
-
Opioids
|
|
Pharmacokinetics and Barbiturates: Introduction and
Overview:
|
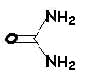
urea |
+ |
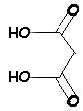
malonic acid |
< > |
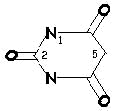
barbituric acid |
-
Particularly advantageous characteristics of thiopental (Pentothal)
(and methohexital (Brevital)) include rapid onset but short duration of action due to
redistribution.
Furthermore, these agents appear relatively safe to use
-
As noted above the primary mechanism for termination of action
of thiopental (Pentothal) and methohexital (Brevital) is
redistribution from the brain to other compartments, as a result
of equilibration. This mechanism is distinct from
metabolism as a means of terminating anesthetic effect.
-
Also, rapid onset is a characteristic of these agents and can
be understood in terms of their chemical properties
-
To understand this aspect we need to consider the molecular
characteristics of these drugs.
-
Barbiturates exists in two molecular forms, and enol and keto
forms.
-
Enol forms for barbiturates are water-soluble at pH
10-11 at 6% sodium carbonate. Therefore, in the
injected form the drug is not initially particularly
lipid-soluble and might not be expected to enter the brain readily.
Here's the structure of enol form for
barbiturate acid:
-
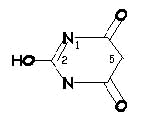
-
Once in circulation, the enol form undergoes a
structural change called "tautomerization" to a keto
form which is more lipid soluble:
-

-
Specific pharmacological characteristics of different
barbiturates are mainly due to chemical substitutions at the
"5" position in the molecule. Hypnotic
properties are influenced by the nature of side chains at
position 5. Duration of action and potency are also
affected by the length of site chains at position 5.
Here are some examples
-
|
Thioamylal:
|
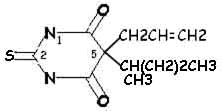
|
-
|
Pentobarbital
(Nembutal) |
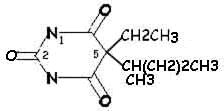
|
-
|
Methohexital (Brevital)
|
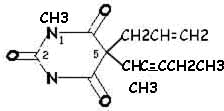
|
-
|
Thiopental (Pentothal) |
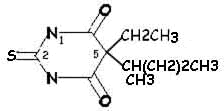
|
Side Chain Differences between Barbituric
Acid Derivatives
|
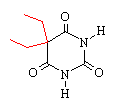
barbital |
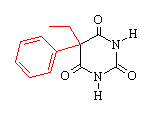
phenobarbital |
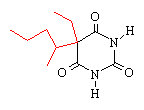
pentobarbital |
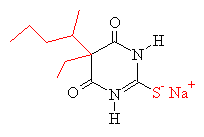
sodium pentothal |
Comparing pentobarbital and thiopental (sodium pentothal), we
note a very subtle structural difference that has a significant
consequence in terms of pharmacokinetic properties. Specifically,
the substitution of sulfur (S) for oxygen (O) is responsible for the
pronounced increased in the partition coefficient (100 times) of
thiopental compared to pentobarbital. The partition coefficient
for thiopental is about 580.
|
1Katzung, B. G. Basic
Principles-Introduction , in Basic and Clinical
Pharmacology, (Katzung, B. G., ed) Appleton-Lange, 1998,
pp 1-33
2Benet, Leslie Z, Kroetz, Deanna
L. and Sheiner, Lewis B The Dynamics of Drug Absorption,
Distribution and Elimination. In, Goodman and Gillman's
The Pharmacologial Basis of Therapeutics,(Hardman, J.G, Limbird, L.E,
Molinoff, P.B., Ruddon, R.W, and Gilman, A.G.,eds) TheMcGraw-Hill Companies, Inc.,1996, pp. 3-27
3Correia, M.A., Drug
Biotransformation. in Basic and Clinical Pharmacology, (Katzung, B. G., ed) Appleton-Lange, 1998, pp 50-61.
4Stoelting, R.K.,
"Pharmacokinetics and Pharmacodynamics of Injected
and Inhaled Drugs", in Pharmacology and Physiology
in Anesthetic Practice, Lippincott-Raven Publishers,
1999, 1-17.
5Dolin, S. J. "Drugs and
pharmacology" in Total Intravenous Anesthesia, pp. 13-35 (Nicholas L.
Padfield, ed), Butterworth Heinemann, Oxford, 2000
|