Medical Pharmacology Chapter 35 Antibacterial Drugs
|
|
|
|
|
|
|
|
|
|
Third Generation Cephalosporin Overview
Mechanism of Action and Resistance
The third-generation cephalosporins are bactericidal beta-lactam antibiotics.
The primary mechanism involves the inhibition of bacterial cell wall synthesis.2
Target Binding
These agents bind to and inhibit specific Penicillin-Binding Proteins (PBPs) which are enzymes like transpeptidases that catalyze the cross-linking of the peptidoglycan polymer.3
Cell Lysis
By preventing the formation of a stable cell wall, the bacteria become subject to osmotic instability.
The binding to PBPs often triggers the release of bacterial autolysins, leading to programmed cell lysis.3
Mechanisms of Resistance
Resistance typically arises through three primary pathways.
(1) Beta-lactamase Production
While third-generation agents are highly resistant to many classic penicillinases, they remain vulnerable to Extended-Spectrum beta-lactamases (ESBLs) and AmpC beta-lactamases.4
(2) PBP Alteration
Modifications in the target PBPs (e.g., PBP2a in MRSA) render these drugs ineffective.
(3) Permeability and Efflux
Mutations in porin channels or the upregulation of efflux pumps in organisms like Pseudomonas aeruginosa can limit drug entry.5
Pharmacological Properties and Pharmacokinetics
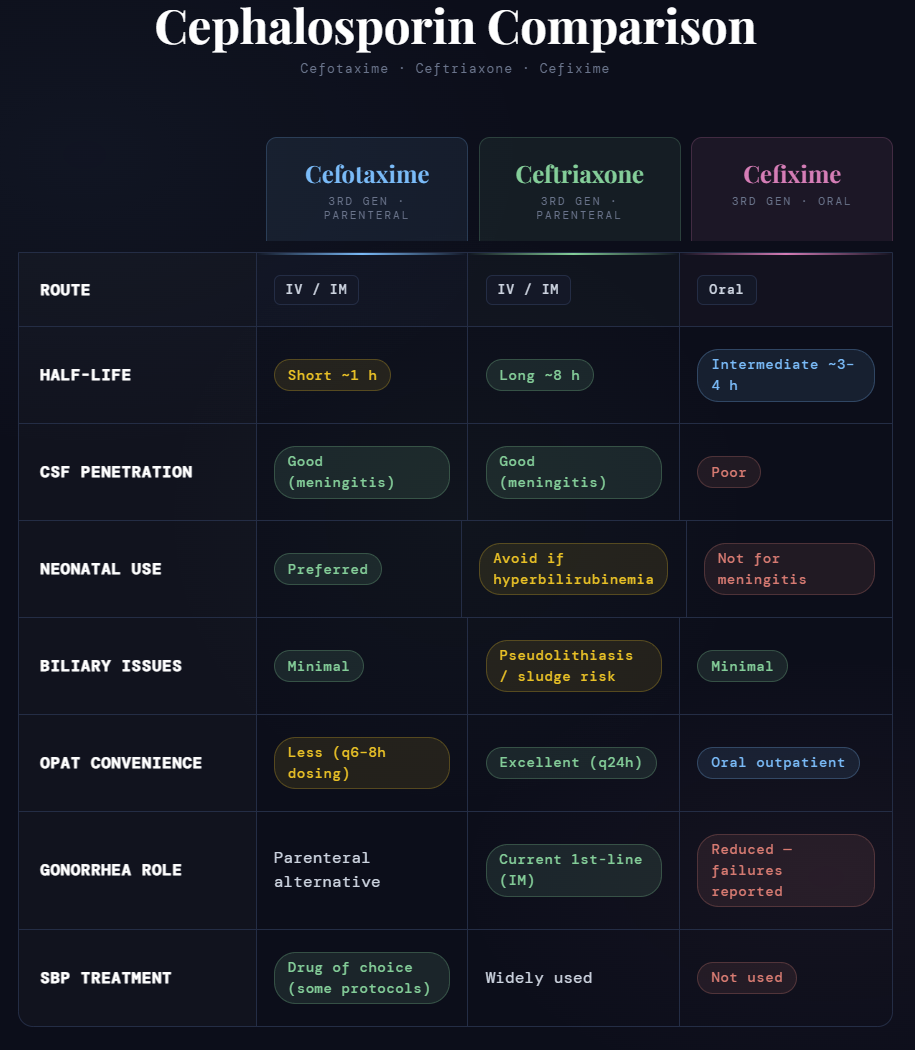
Cefotaxime (Parenteral)
Overview and Pharmacological Properties
Cefotaxime is a broad-spectrum parenteral beta-lactam that serves as a cornerstone of hospital-based therapy.
One of its most distinctive features is its hepatic metabolism into desacetylcefotaxime.
This metabolite is microbiologically active and acts synergistically with the parent drug, particularly against Haemophilus influenzae.6
With a relatively short half-life of approximately 1 hour, it typically requires dosing every 6 to 8 hours for systemic infections.
Cefotaxime is primarily excreted through the kidneys via glomerular filtration and tubular secretion.7
Mechanism of Action
Cefotaxime exerts its bactericidal effect by binding to and inactivating Penicillin-Binding Proteins (PBPs), the transpeptidases, located on the inner bacterial cell membrane.
This binding inhibits the final transpeptidation step of peptidoglycan synthesis, causing the cell wall to lose structural integrity.
Resulting osmotic instability, combined with the activation of bacterial autolysins, leads to rapid cell lysis.
Synergy between the parent drug and its desacetyl metabolite is an important pharmacological advantage.8
Clinical Uses
Neonatal Sepsis
Neonatal sepsis is a systemic, life-threatening infection in infants less than 28 days old, often caused by E. coli or Group B Streptococcus.
Cefotaxime is a recommended agent because it provides robust Gram-negative coverage without displacing bilirubin from albumin.9
Bacterial Meningitis
Bacterial meningitis is a life-threatening inflammation of the protective membranes (meninges) covering the brain and spinal cord.
Cefotaxime is effective in achieving therapeutic concentrations in the cerebrospinal fluid (CSF) during active inflammation.10
Intra-abdominal Infections
These infections, such as peritonitis, typically follow a breach in the gastrointestinal tract.
Cefotaxime targets the aerobic Gram-negative bacilli involved, often paired with metronidazole to provide anaerobic coverage.
Adverse Effects
Common side effects include hypersensitivity reactions like rashes and local injection site reactions.
Because of its renal clearance, high doses in patients with renal failure can lead to neurotoxicity, including seizures.
Case Study: The Febrile Neonate
Presentation
A 10-day-old infant is brought to the emergency department with poor feeding, lethargy, and a temperature of 39.1°C (102.4°F).
Discussion
![]() The
clinical priority is empiric coverage for neonatal sepsis.
The
clinical priority is empiric coverage for neonatal sepsis.
The chosen regimen is ampicillin plus cefotaxime.
This selection is important because, unlike ceftriaxone, cefotaxime does not compete for bilirubin binding sites on albumin.
![]() In a
neonate with an immature blood-brain barrier, using cefotaxime
avoids the very serious risk of bilirubin-induced encephalopathy
(kernicterus).13
In a
neonate with an immature blood-brain barrier, using cefotaxime
avoids the very serious risk of bilirubin-induced encephalopathy
(kernicterus).13
Practice Question
Question
What is the primary pharmacological reason cefotaxime is used instead of ceftriaxone in a 2-week-old infant?
Answer
Cefotaxime does not displace bilirubin from albumin, reducing the risk of kernicterus.14
Ceftriaxone (Parenteral)
Overview and Pharmacological Properties
Ceftriaxone is characterized by a long elimination half-life of 6 to 9 hours.
The long half-life allows for convenient once-daily dosing in most clinical settings.
Ceftriaxone utilizes a dual elimination pathway, with approximately 60% excreted renally and 40% through the biliary system.15
Mechanism of Action
Ceftriaxone acts by binding to PBPs to inhibit bacterial cell wall synthesis.
Resistance in organisms like Pseudomonas aeruginosa is often driven by OprD porin loss (restricting drug entry) and MexAB-OprM efflux upregulation (active outward translocation of the drug).5
Clinical Uses
Gonorrhea
A sexually transmitted infection caused by Neisseria gonorrhoeae that can lead to pelvic inflammatory disease.
A single intramuscular dose of ceftriaxone is the primary treatment of choice.16
Infective Endocarditis17
This condition represents a serious infection of the heart valves.
Ceftriaxone is often used for stable patients finishing a course of IV antibiotics at home (OPAT) because its once-daily dosing improves compliance.
Lyme Disease
Ceftriaxone is the preferred treatment for advanced stages, such as Lyme meningitis or carditis, due to its excellent tissue penetration.18
Adverse Effects
Biliary Sludging
High biliary concentrations can lead to the formation of calcium-ceftriaxone precipitates in the gallbladder, manifesting as "pseudolithiasis."19
Contraindications
Ceftriaxone is strictly contraindicated in neonates and with calcium-containing IV fluids.20
Cefixime (Oral)
Overview and Pharmacological Properties
Cefixime provides a critical oral option within the third generation.
Cefixime has an oral bioavailability of roughly 40% to 50% and a half-life of 3 to 4 hours. and is cleared by both renal and biliary mechanisms.21
Mechanism of Action
Like the parenteral agents, cefixime inhibits cell wall synthesis by binding to PBPs.
However, cefixime has a significantly lower affinity for the PBPs of Staphylococcus aureus, making it an inappropriate choice for staphylococcal infections.22
Clinical Uses23
Uncomplicated Urinary Tract Infections (UTIs)
A UTI is an infection of the bladder (cystitis) or kidneys (pyelonephritis).
Cefixime is an excellent oral choice for UTIs caused by E. coli or Proteus mirabilis.
Acute Exacerbation of Chronic Bronchitis
This condition involves a sudden worsening of airway inflammation in patients with chronic lung disease.
 |
February, 2026
|
|
This Web-based pharmacology and disease-based integrated teaching site is based on reference materials, that are believed reliable and consistent with standards accepted at the time of development. Possibility of human error and on-going research and development in medical sciences do not allow assurance that the information contained herein is in every respect accurate or complete. Users should confirm the information contained herein with other sources. This site should only be considered as a teaching aid for undergraduate and graduate biomedical education and is intended only as a teaching site. Information contained here should not be used for patient management and should not be used as a substitute for consultation with practicing medical professionals. Users of this website should check the product information sheet included in the package of any drug they plan to administer to be certain that the information contained in this site is accurate and that changes have not been made in the recommended dose or in the contraindications for administration. Advertisements that appear on this site are not reviewed for content accuracy and it is the responsibility of users of this website to make individual assessments concerning this information. Medical or other information thus obtained should not be used as a substitute for consultation with practicing medical or scientific or other professionals. |