Medical Pharmacology Chapter 35 Antibacterial Drugs
Third Generation Cephalosporins in More Detail
Introduction
Third‑generation cephalosporins—cefotaxime, ceftriaxone, and cefixime—are broad‑spectrum β‑lactam antibiotics central to modern management of serious Gram‑negative and many community‑acquired infections, especially in CNS, respiratory, abdominal, genitourinary, and systemic disease.
These drugs share a common mechanism but differ markedly in pharmacokinetics, formulation, and safety, which drives their distinct roles in clinical infectious‑diseases practice.2,3,6,7
Mechanism of action and resistance3,4,5
Cephalosporins inhibit bacterial cell wall synthesis by binding to penicillin‑binding proteins (PBPs) and blocking the transpeptidation and cross‑linking of peptidoglycan, producing structurally weak cell walls and osmotic lysis of susceptible bacteria.
Bactericidal activity is time‑dependent
Clinical efficacy correlates with the fraction of the dosing interval for which free drug concentrations exceed the MIC (fT>MIC), which underlies the frequent dosing of cefotaxime and high single doses of ceftriaxone.
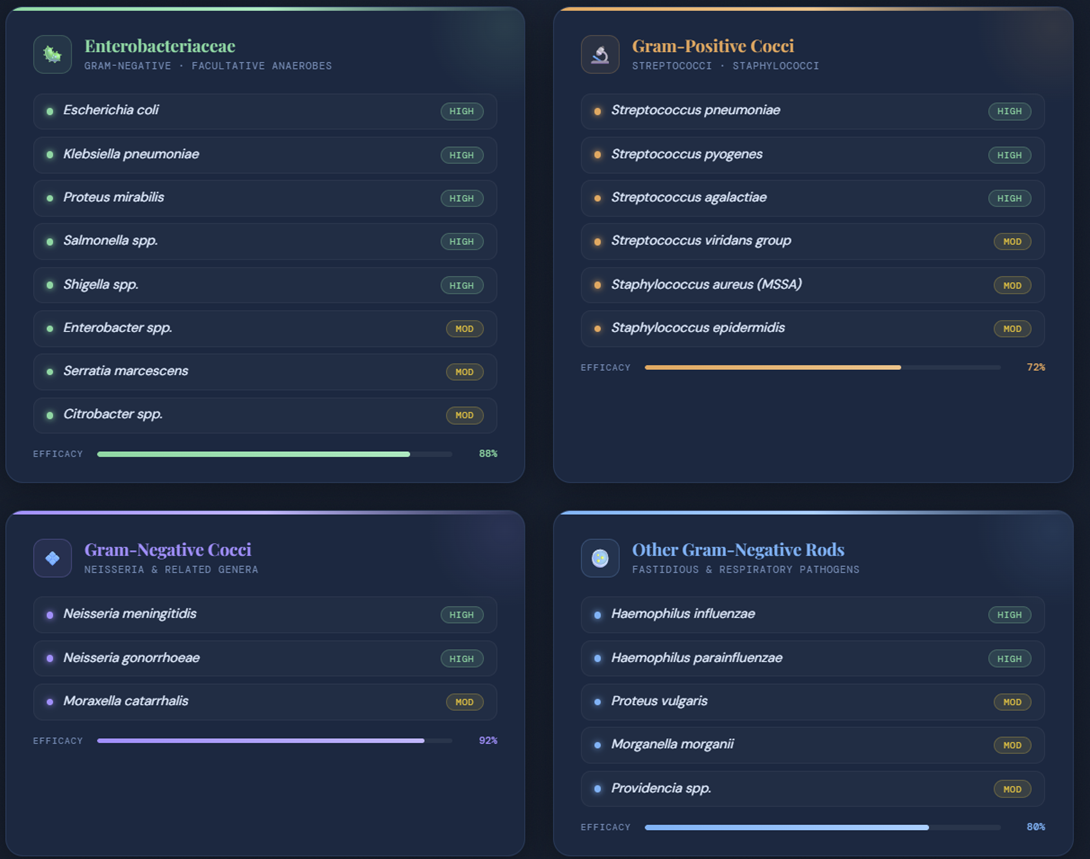
Third‑generation cephalosporins are relatively stable to many
plasmid‑encoded β‑lactamases and have enhanced activity versus
Enterobacterales, Neisseria, and Haemophilus
spp., but they are hydrolyzed by
![]() Extended-Spectrum Beta-Lactamases
(ESBLs) (e.g., CTX‑M family) and many AmpC enzymes.
Extended-Spectrum Beta-Lactamases
(ESBLs) (e.g., CTX‑M family) and many AmpC enzymes.
![]() Resistance
develops as result of:2,3,8
Resistance
develops as result of:2,3,8
ESBLs and AmpC β‑lactamases that efficiently hydrolyze cefotaxime, ceftriaxone, and cefixime, often leaving carbapenems or newer β‑lactam/β‑lactamase‑inhibitor combinations as preferred options.2,3,8
Changes in Penicillin-Binding Proteins (PBPs) in Streptococcus pneumoniae and Neisseria gonorrhoeae, which increase MICs and have contributed to cefixime clinical failures in gonorrhea.9,10,11,12
Decreased outer‑membrane permeability and upregulated efflux pumps in Gram‑negative bacilli that act synergistically with β‑lactamase production.2,3
These mechanisms have resulted in major guideline changes, including removal of cefixime from first‑line gonorrhea treatment in favor of higher‑dose IM ceftriaxone.10,13,14
|
 |
 |
Mechanism and pharmacological profile
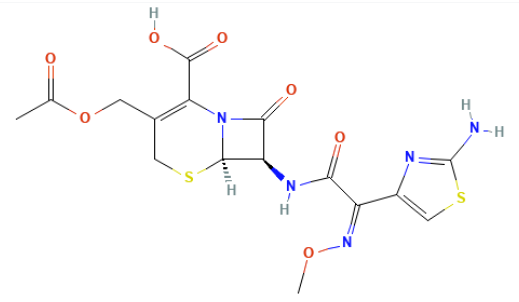
![]() Cefotaxime
is a parenteral third‑generation cephalosporin active
against Enterobacterales (E. coli,
Klebsiella, Proteus, some Enterobacter),
Neisseria, H. influenzae, and many
streptococci including S. pneumoniae;
Cefotaxime
is a parenteral third‑generation cephalosporin active
against Enterobacterales (E. coli,
Klebsiella, Proteus, some Enterobacter),
Neisseria, H. influenzae, and many
streptococci including S. pneumoniae;
Cefotaxime exhibits weaker antistaphylococcal activity compared to first‑generation cephalosporins and lacks significant anaerobic and Pseudomonas coverage.
Cefotaxime, similar to other β‑lactams, binds multiple PBPs, with high affinity for those essential to cell wall cross‑linking in Gram‑negative bacilli.2,4,15,16
 |
 |
 |
Principal pharmacokinetic characteristics2, 5,17
Administration and distribution: Given IV or IM (no oral formulation), cefotaxime distributes rapidly into extracellular fluids; adult volumes of distribution are approximately 20–27 L, indicating extensive tissue penetration.
Cefotaxime penetrates well into lung, pleural, peritoneal, bone, synovial fluid, and inflamed meninges.2,5,16,17
|
|
|
In infants receiving 50 mg/kg every 6 h, mean serum concentrations 15 minutes after infusion were around 120 μg/mL, with CSF concentrations around 6 μg/mL and mean CSF:serum ratios near 0.29 in inflamed meninges.
This drug concentration is deemed sufficient to exceed MICs for N. meningitidis, H. influenzae, and most S. pneumoniae strains.5,18
Metabolism and Elimination15,17
Cefotaxime is partly metabolized to desacetylcefotaxime, an active metabolite with a longer half‑life (~2 h) and complementary antibacterial activity.
Parent drug and metabolite are primarily renally eliminated;.
Total half‑life in adults with normal renal function is ~0.8–1 h for cefotaxime and ~2 h for desacetylcefotaxime,.
Given these half-lives, dosing is likely required every 6–8 h in serious infections.
Significant renal impairment requires dose reduction or prolonged dosing intervals to avoid accumulation and neurotoxicity.15,17
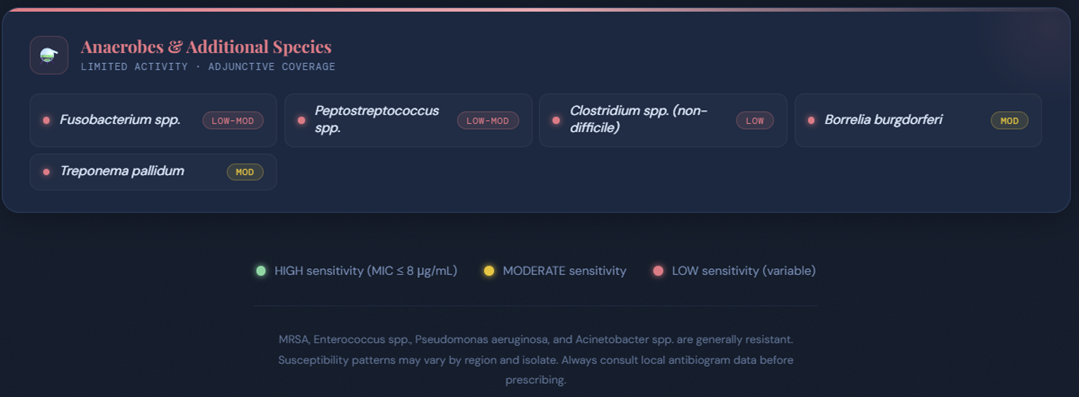
Cefotaxime: Therapeutic Uses (detailed)
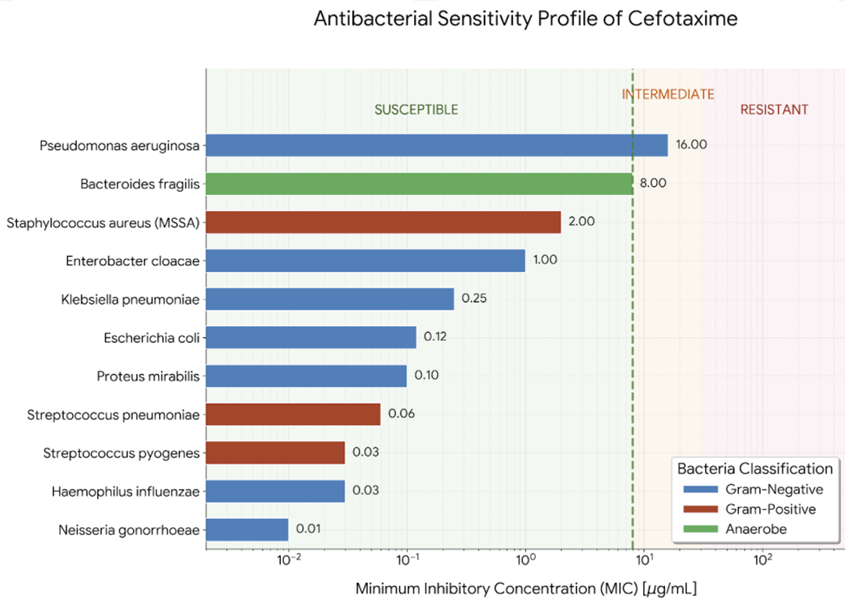
Cefotaxime is a mainstay for hospitalized patients with serious infections when Pseudomonas and ESBLs are not major concerns.15,16,19
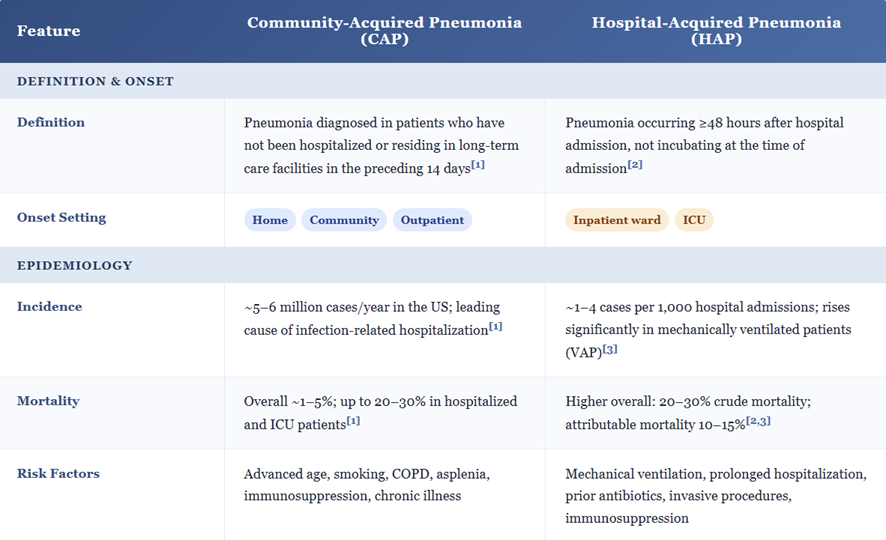
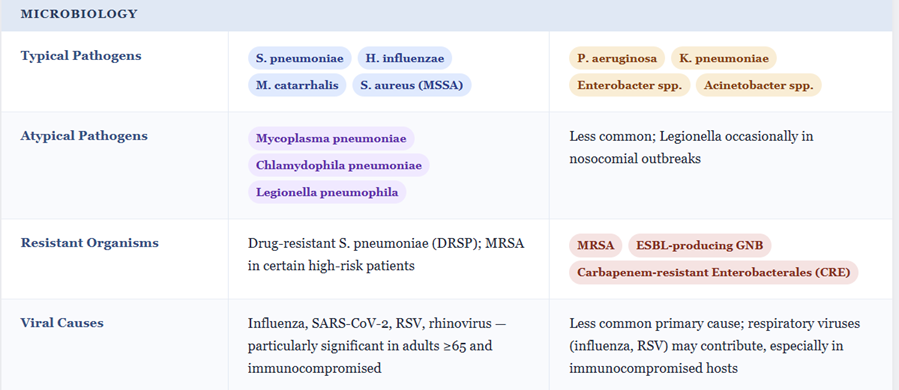
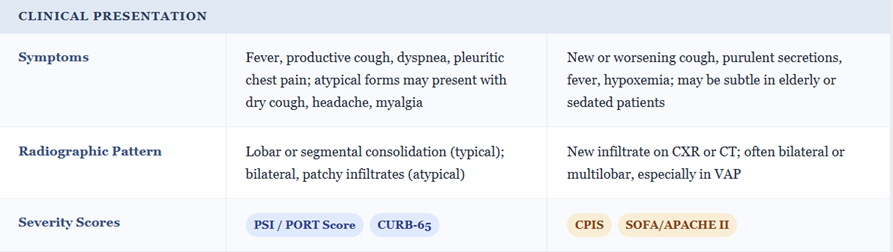
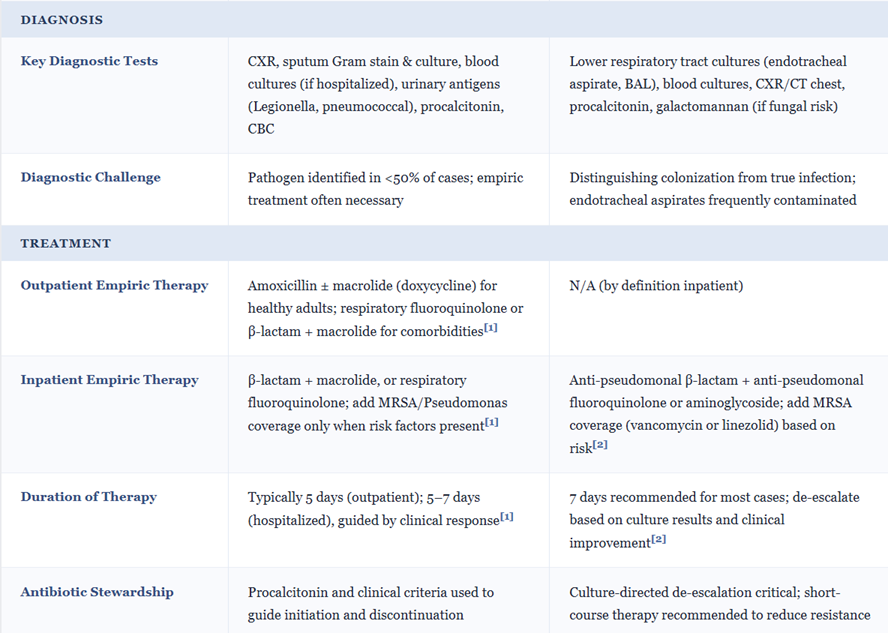
![]() Community‑acquired and hospital‑acquired pneumonia20,21,22
Community‑acquired and hospital‑acquired pneumonia20,21,22
In Community Acquired Pneumonia (CAP) requiring hospitalization, cefotaxime (1–2 g IV q8h) is recommended by ATS/IDSA as one of the β‑lactam options combined with a macrolide or doxycycline agent. The β-lactam targets S. pneumoniae, H. influenzae, and Enterobacterales while the other drug covers atypicals.
In early Hospital Acquired Pneumonia (HAP) without high Multi-Drug Resistance (MDR) risk, Cefotaxime can be part of empiric therapy, but in later‑onset or high‑risk HAP, broader anti‑pseudomonal β‑lactams are preferred.
 |
 |
 |
 |
 |
|
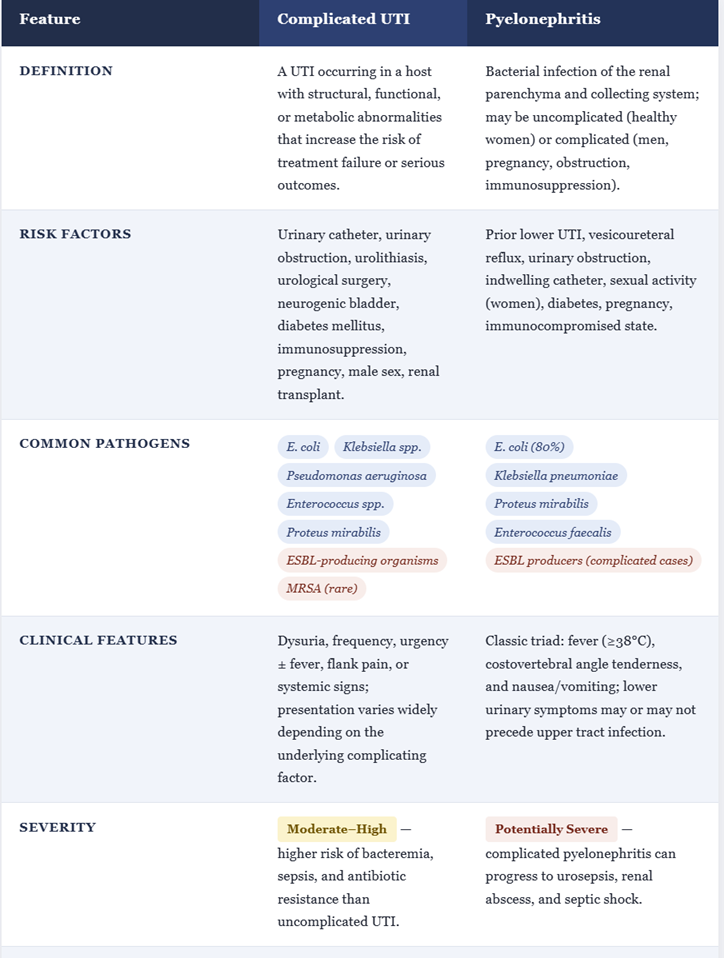
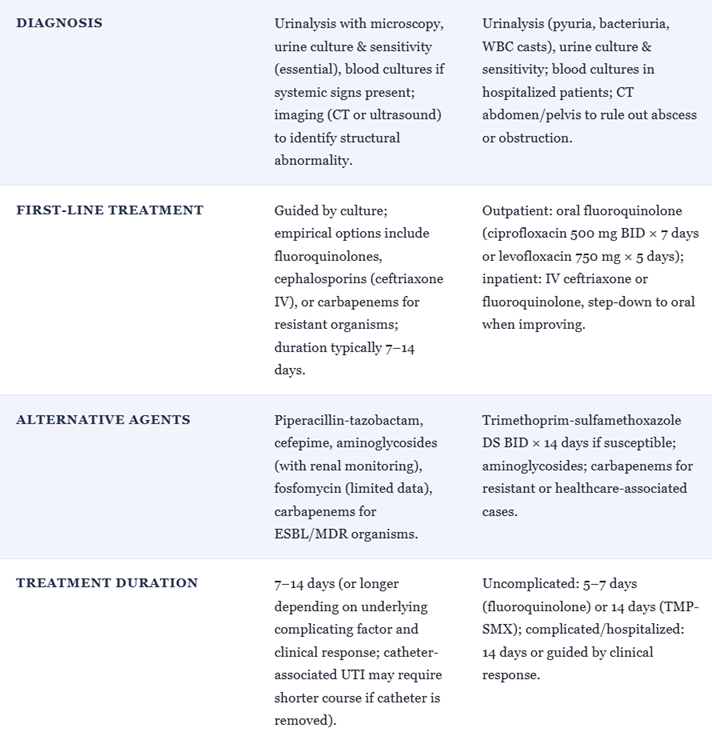
![]() Complicated Urinary Tract Infections and Pyelonephritis16,19
Complicated Urinary Tract Infections and Pyelonephritis16,19
For hospitalized pyelonephritis or complicated UTIs due to susceptible Enterobacterales, cefotaxime achieves high serum and urinary concentrations and is effective where fluoroquinolone resistance is problematic.
Cefotaxime is often used as initial IV therapy in the Emergency Department or inpatient settings, followed by oral step‑down once the patient is stable and susceptibilities permit.
 |
 |
 |
|
February, 2026
|
|
This Web-based pharmacology and disease-based integrated teaching site is based on reference materials, that are believed reliable and consistent with standards accepted at the time of development. Possibility of human error and on-going research and development in medical sciences do not allow assurance that the information contained herein is in every respect accurate or complete. Users should confirm the information contained herein with other sources. This site should only be considered as a teaching aid for undergraduate and graduate biomedical education and is intended only as a teaching site. Information contained here should not be used for patient management and should not be used as a substitute for consultation with practicing medical professionals. Users of this website should check the product information sheet included in the package of any drug they plan to administer to be certain that the information contained in this site is accurate and that changes have not been made in the recommended dose or in the contraindications for administration. Advertisements that appear on this site are not reviewed for content accuracy and it is the responsibility of users of this website to make individual assessments concerning this information. Medical or other information thus obtained should not be used as a substitute for consultation with practicing medical or scientific or other professionals. |