 |
-
Neurocognitive Impairment in Bipolar Disorder
-
Cognitive Profile in BD and the Role of Psychosis
-
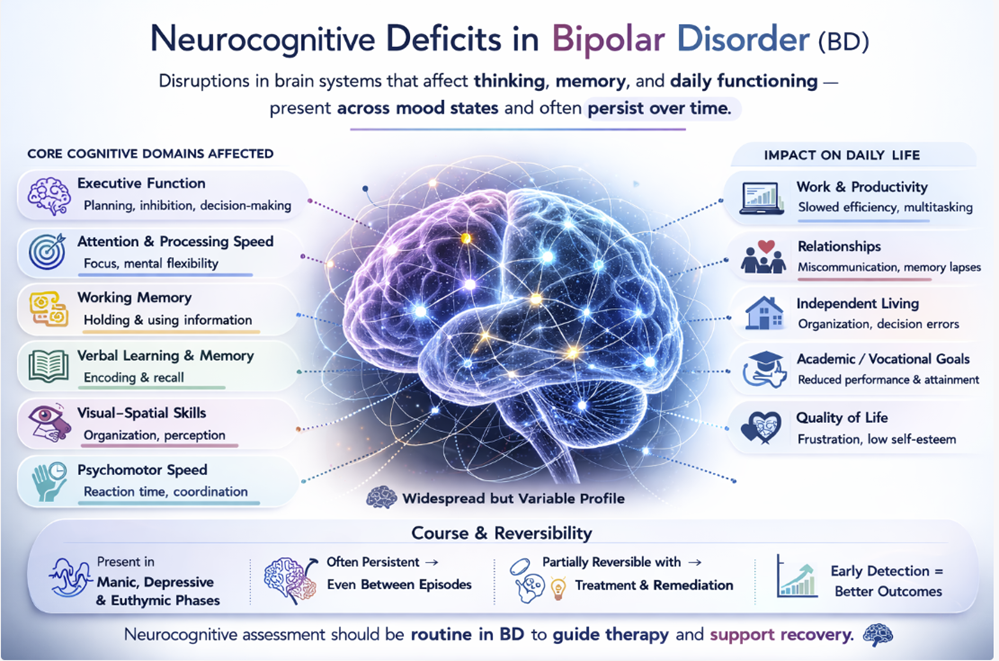
Neurocognitive impairment in bipolar disorder has gained increasing recognition as an important illness dimension, moving the field beyond the earlier view that cognitive difficulties were purely state-dependent phenomena related to acute mood episodes. Meta-analyses of euthymic BD patients demonstrate consistent moderate deficits in verbal memory, executive function, attention, and processing speed, with typical effect sizes of 0.5–0.8 standard deviations below healthy controls [33, 34]. These effect sizes are substantially smaller than those observed in schizophrenia but are nonetheless clinically meaningful and are associated with functional impairment even in the absence of active mood symptoms. Arts and colleagues [33] found that unaffected relatives of BD patients showed intermediate (though significant) deficits, supporting a genetic contribution to these impairments, paralleling similar observations in schizophrenia.
-
-
 The presence of psychotic features within BD significantly modifies the
cognitive picture.
The presence of psychotic features within BD significantly modifies the
cognitive picture.
-
Bipolar patients with a history of psychosis consistently perform worse on neuropsychological testing than those without, and their profiles more closely resemble those of schizophrenia patients.35
-
Specific domain comparisons show that BD with psychosis approximates schizophrenia in verbal memory and processing speed impairment, while schizophrenia patients maintain an additional disadvantage particularly in working memory and social cognition.36
-
-
-
Cognitive Spectrum and Diagnostic Implications
-
Cognitive deficits appear to increase across a spectrum from unipolar depression, through BD without psychosis, through BD with psychosis, through schizoaffective disorder, to schizophrenia exhibiting a gradient-like severity increments rather than clean categorical breaks.37
-
This graded relationship is one of the most compelling pieces of evidence for a psychosis continuum,
-
-
Neuropsychological testing alone cannot reliably differentiate psychotic BD from schizophrenia, but a patient with pervasive, stable cognitive deficits across multiple domains present from before illness onset and without clear relationship to affective state, strongly suggests a schizophrenia spectrum diagnosis28,36
-
-
-
-
Functional Outcomes in Schizophrenia
-
 Schizophrenia ranks among the most functionally disabling conditions
in all of medicine.
Schizophrenia ranks among the most functionally disabling conditions
in all of medicine.
-
The Global Burden of Disease studies consistently place schizophrenia among the top ten causes of years lived with disability worldwide, despite its relatively low prevalence (approximately 0.5–1%), reflecting the profound impairment in occupational, social, and self-care functioning that characterises the disorder.38
-
Unemployment rates of 80–90% are documented in many Western countries, far exceeding those of any other psychiatric condition.
-
 Cognitive
impairment, particularly in processing speed, verbal
memory, and social cognition is a stronger predictor of
functional outcome than positive symptom severity in
most longitudinal analyses.39
Cognitive
impairment, particularly in processing speed, verbal
memory, and social cognition is a stronger predictor of
functional outcome than positive symptom severity in
most longitudinal analyses.39
-
 Negative symptoms directly impair motivation, hedonic
capacity, and goal-directed behaviour, often resulting
in poor work performance and social withdrawal.
Negative symptoms directly impair motivation, hedonic
capacity, and goal-directed behaviour, often resulting
in poor work performance and social withdrawal.
-
-
-
-
Social functioning in schizophrenia is compromised at multiple levels:
-
Social cognition deficits impair the ability to accurately read social cues and mentalize (where mentalizing represents cognitive and emotional process of interpreting behavior).
-
Negative symptoms reduce motivation for social engagement.
-
Paranoid ideation creates avoidance of social situations
-
Institutionalisation and stigma further erode social capital over time.27,40
-
-
 Patients
with schizophrenia have markedly reduced rates of marriage and
stable partnership, reduced social network size, and high rates
of social isolation compared to both general population controls
and patients with mood disorders.
Patients
with schizophrenia have markedly reduced rates of marriage and
stable partnership, reduced social network size, and high rates
of social isolation compared to both general population controls
and patients with mood disorders.
-
-
Functional Outcomes in Psychotic Bipolar Disorder (BD)
-
Functional outcomes in BD are substantially better, on average, than in schizophrenia, but are worse than commonly assumed, especially in patients with psychotic features.
-
The view of BD as an episodic condition with full interepisodic recovery has been progressively revised by longitudinal data showing that between episode functional impairment is common, particularly after multiple episodes and in those with psychotic features or comorbid substance use.20,40
-
The STEP-BD study found that among patients achieving syndromal recovery, substantial proportions continued to report functional impairment in occupational, social, and recreational domains — a finding labelled 'functional non-recovery' [20].
-
STEP-BD: Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). https://www.nimh.nih.gov/funding/clinical-research/practical/step-bd
-
-
Quality of life assessments in BD with psychosis consistently show lower Quality of Live (OoL) relative to BD without psychosis, and QoL domains related to social functioning and mental health are the most severely affected, though patients with BD generally report higher QoL than patients with schizophrenia on most instruments.40
-
-
-
-
-
-
Familial Aspects: Heritability, Aggregation, and Risk Transmission
-
-
 Schizophrenia
is among the most heritable of all psychiatric disorders.
Schizophrenia
is among the most heritable of all psychiatric disorders.
-
Twin studies have consistently documented monozygotic (MZ) concordance rates of approximately 41–65% and dizygotic (DZ) concordance rates of approximately 0–28%, yielding heritability estimates in the range of 64–80% [41].
-
Adoption studies converge on the same conclusion:
-
 Children
of parents with schizophrenia who are raised by adoptive
parents without schizophrenia retain their elevated genetic
risk, confirming that familial aggregation reflects shared
genes rather than shared environment.
Children
of parents with schizophrenia who are raised by adoptive
parents without schizophrenia retain their elevated genetic
risk, confirming that familial aggregation reflects shared
genes rather than shared environment.
-
First-degree relatives of patients with schizophrenia have an approximately 10-fold elevated lifetime risk (~6–13%) relative to the general population, with second-degree relatives showing intermediate elevated risk (~3–4%), consistent with polygenic inheritance.42
-
-
-
 This
familial predispositions extends not only to schizophrenia but
to a broader schizophrenia spectrum including schizotypal
personality disorder and schizoaffective disorder.
This
familial predispositions extends not only to schizophrenia but
to a broader schizophrenia spectrum including schizotypal
personality disorder and schizoaffective disorder.
-
-
-
Heritability of Bipolar Disorder
-
Bipolar disorder is also highly heritable, with twin studies yielding MZ concordance rates of approximately 40–70% for bipolar I disorder and heritability estimates ranging from 59–85%.
-
McGuffin and colleagues conducted one of the most methodologically careful twin studies of BD heritability, documenting heritability of approximately 85% for bipolar I disorder using structured diagnostic interviews.43
-
First-degree relatives of bipolar I probands have a lifetime risk of approximately 5–15% for BD.
-
A key observation in the familial epidemiology of BD is that the transmitted liability is not simply to a specific DSM category,
-
Families of BD probands (a person serving as the starting point for the genetic study of a family) show elevated rates of BD I, BD II, cyclothymia, and recurrent unipolar depression
-
These observations suggested that what is transmitted is a broader mood dysregulation spectrum.44
-
-
-
-
-
-
Cross-Disorder Familial Co-Aggregation
-
Perhaps the most direct evidence for shared familial liability between schizophrenia and BD comes from epidemiological studies of familial co-aggregation.
-
The Swedish national register study by Lichtenstein and colleagues.2, using over nine million individuals, demonstrated that first-degree relatives of patients with schizophrenia, had elevated risks not only of schizophrenia but also of bipolar disorder, and vice versa.
-
Also, relatives of BD probands showed elevated risks of both BD and schizophrenia. [(probands: "a person serving as the starting point for the genetic study of a family (used especially in medicine and psychiatry)"]
-
Half-siblings shared less of this cross-disorder familial risk than full siblings, confirming that the co-aggregation reflects genetic sharing.
-
-
 This
landmark study estimated the genetic correlation between schizophrenia
and bipolar disorder at approximately 0.60, meaning the majority, though
not all, of the genetic liability to one condition was shared with the
other.
This
landmark study estimated the genetic correlation between schizophrenia
and bipolar disorder at approximately 0.60, meaning the majority, though
not all, of the genetic liability to one condition was shared with the
other. -
Subsequent Genome-Wide Association Study (GWAS)-based analyses have refined this estimate to 0.60–0.70.3,45
-
-
-
-