Medical Pharmacology Chapter 16: Pharmacology of Antipsychotics Drugs
First Generation Antipsychotic Medications
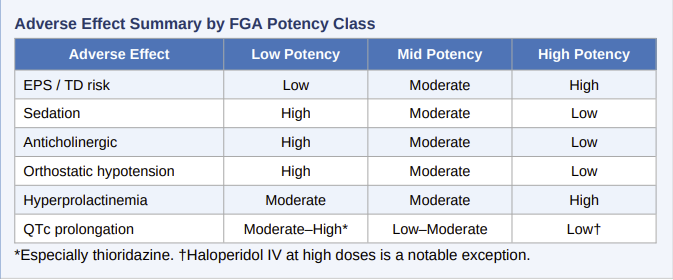
![]() Blockade
of muscarinic M1 receptors produces a predictable anticholinergic
adverse effect that is especially pronounced with low-potency FGAs such as
chlorpromazine and thioridazine.
Blockade
of muscarinic M1 receptors produces a predictable anticholinergic
adverse effect that is especially pronounced with low-potency FGAs such as
chlorpromazine and thioridazine.
Common peripheral effects include dry mouth, constipation, urinary retention, blurred vision, and decreased sweating (with risk of heat intolerance).
Central anticholinergic effects, especially in elderly patients include memory impairment, confusion, and delirium.
Concurrent use of anticholinergic agents prescribed to manage Extrapyramidal Symptoms (EPS) such as benztropine or trihexyphenidyl, substantially increase this risk.
Sedation and Orthostatic Hypotension
H1 antihistamine activity produces sedation that may be clinically useful in the acute setting (particularly for agitation) but is a significant burden for outpatient maintenance.
![]() Low-potency agents such as chlorpromazine are among the most sedating
antipsychotics available.
Low-potency agents such as chlorpromazine are among the most sedating
antipsychotics available.
α-1 adrenergic blockade produces orthostatic hypotension, most pronounced upon initial dosing.
Falls and related injuries, particularly in elderly patients, represent a serious consequence.
![]() Blood
pressure monitoring in the first weeks of treatment is advisable, especially
in patients with cardiovascular disease or those on concurrent
antihypertensives.
Blood
pressure monitoring in the first weeks of treatment is advisable, especially
in patients with cardiovascular disease or those on concurrent
antihypertensives.
Endocrine and Metabolic Effects
Tuberoinfundibular D2 blockade releases dopamine's tonic inhibition of pituitary lactotrophs, resulting in hyperprolactinemia.
Clinical consequences include galactorrhea, amenorrhea, and sexual dysfunction in women, and gynecomastia, erectile dysfunction, and reduced libido in men.
Prolonged hyperprolactinemia may reduce bone density.
First Generation Antipsychotics (FGAs) as a class cause significantly less metabolic disruption (weight gain, dyslipidemia, glucose dysregulation) than many SGAs, particularly olanzapine and clozapine, a clinical consideration when metabolic comorbidities are a concern.
Cardiovascular and Other Effects
![]() QTc
prolongation is a risk with several FGAs, most notably thioridazine and
mesoridazine, which are associated with a dose-dependent risk of torsades de
pointes and sudden cardiac death.
QTc
prolongation is a risk with several FGAs, most notably thioridazine and
mesoridazine, which are associated with a dose-dependent risk of torsades de
pointes and sudden cardiac death.
Thioridazine carries a black-box warning and is now restricted to refractory cases where other agents have failed.
Haloperidol, while generally considered safer in this regard, has been associated with QTc prolongation at high intravenous doses.
![]() Baseline
ECG, electrolyte monitoring, and caution with concurrent QT-prolonging
agents are essential.
Baseline
ECG, electrolyte monitoring, and caution with concurrent QT-prolonging
agents are essential.
Photosensitivity reactions (particularly with chlorpromazine), pigmentary retinopathy (thioridazine at doses >800 mg/day), cholestatic jaundice, and agranulocytosis (rare, but observed with low-potency phenothiazines) are additional adverse effects meriting clinical attention.
 |
|
DISCLAIMER
|