|
|
Chapter 3: General Principles: Pharmacodynamics
Introduction: The Slow-Signaling Receptor Families
The prior section covered the two fastest signal transducers in pharmacology:
(1) G-protein-coupled receptors (GPCRs) (seconds to minutes) and
(2) Ligand-gated ion channels (milliseconds).
This section considers two remaining receptor superfamilies, which operate on different timescales and through different mechanisms:
(1) Enzyme-linked receptors (minutes to hours) and
(2) Nuclear receptors (hours to days).
The difference in speed is due to a difference in receptor function.
GPCRs and LGICs regulate rapid physiological adjustments: heart rate, neurotransmission, vascular tone, muscle contraction.
Enzyme-linked receptors regulate cell growth, proliferation, differentiation, and survival and these responses that are inherently slower and longer-lasting.
Nuclear receptors regulate gene transcription directly thus producing effects that remain long after the drug has been eliminated.
Understanding these receptor families at a mechanistic level provides useful clinical insights.
Enzyme-linked receptors are the targets of a rapidly expanding class of targeted cancer drugs and biologic immunomodulators.
Nuclear receptors are the targets of glucocorticoids, sex hormones, thyroid hormones, vitamin D, and retinoids, representing agents in drug classes frequently presecribed.
The adverse effect profiles of all these drugs are closely linked to their receptor mechanisms.1,2
Receptor Tyrosine Kinases: Structure and Activation
 |
Receptor tyrosine kinases (RTKs) are single-pass transmembrane proteins in contrast to the seven-pass architecture of GPCRs.
RTKs cross the cell membrane only once.
Receptor tyrosine kinases share a conserved domain organization consisting of
(1) Lrge extracellular ligand-binding domain
(2) Single hydrophobic transmembrane helix, and an
(3) Intracellular domain containing the catalytic tyrosine kinase region.3
RTKs are activated by peptide and protein ligands such as growth factors, cytokines, and hormones that cannot cross the cell membrane and must signal through surface receptors.
Clinically important RTK families and their ligands include:
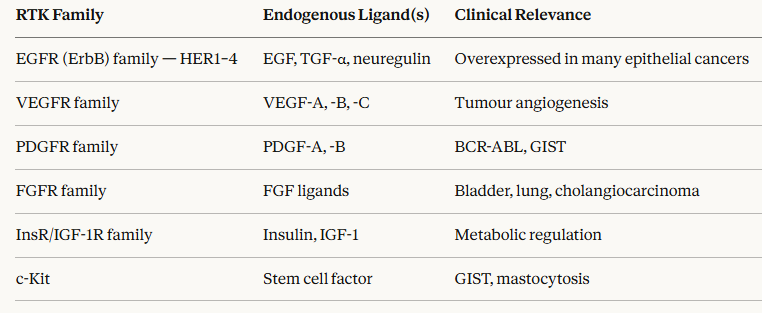 |
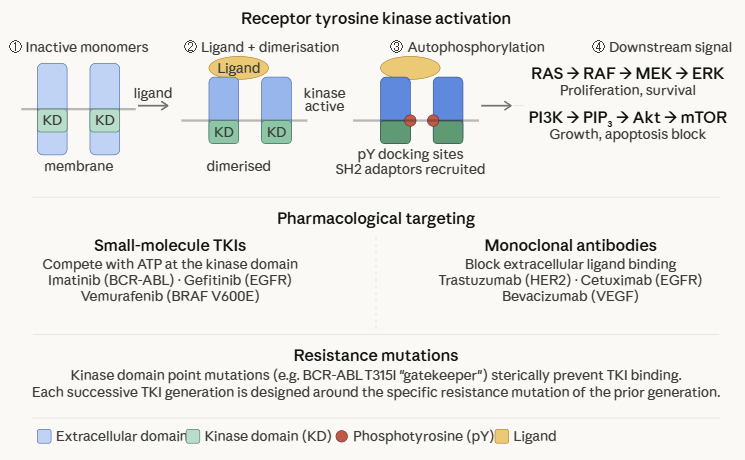
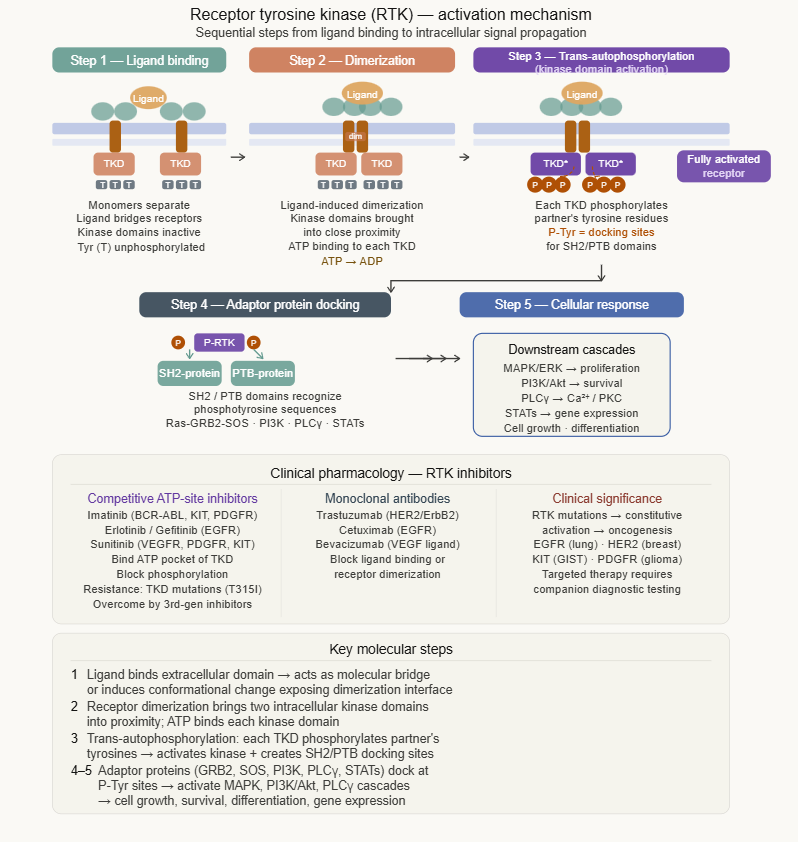
The activation mechanism proceeds through a well-characterized sequence.4,5
Most RTKs exist as monomers in the absence of ligand.
Ligand binding induces receptor dimerization with the two receptor monomers are brought together in the plane of the membrane, either directly (ligand acts as a bridge) or through ligand-induced conformational changes that expose dimerization interfaces.
Dimerization brings the two intracellular kinase domains into proximity.
Each kinase domain then phosphorylates specific tyrosine residues on its partner, a process called trans-autophosphorylation.
Phosphorylation of key tyrosine residues in the activation loop of the kinase domain stabilizes the active kinase conformation, increasing catalytic activity.
Additional phosphorylated tyrosines on the intracellular domain serve as docking sites for intracellular signalling proteins containing SH2 (Src homology 2) or PTB (phosphotyrosine binding) domains, proteins that recognise and bind specific phosphotyrosine-containing sequences.4
 |
RTK Downstream Signaling Cascades
The intracellular signaling networks activated by receptor tyrosine kinases (RTKs) are among the most clinically important in all of pharmacology, because their dysregulation, through mutation, amplification, or fusion, drives a large proportion of human cancers.
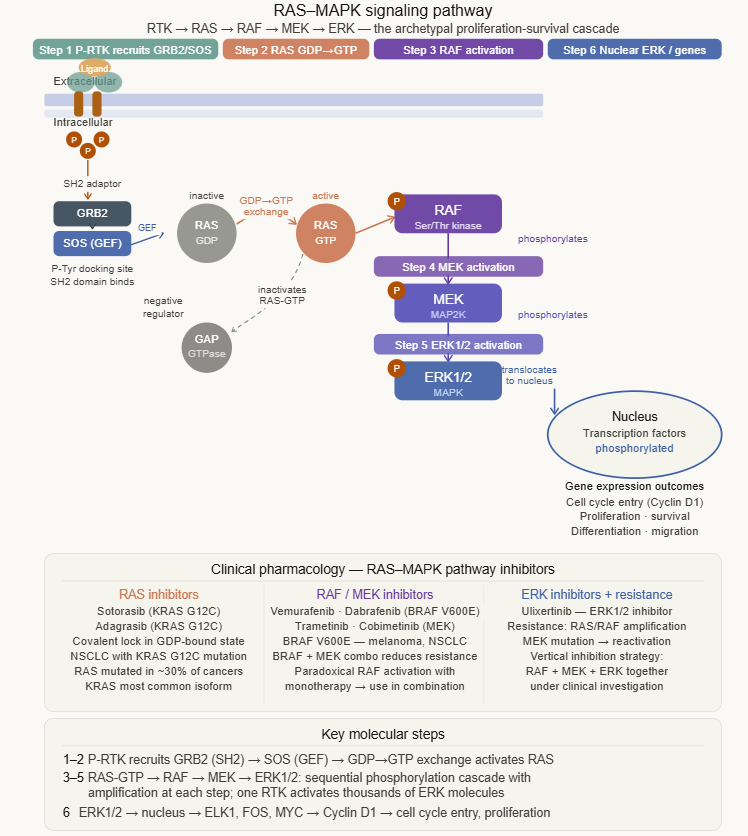
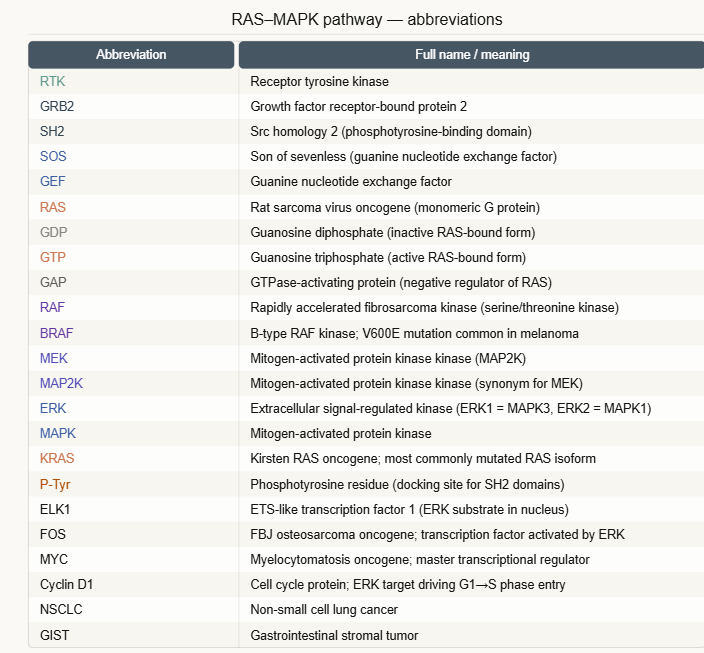
The RAS–MAPK pathway is the archetypal proliferation-survival cascade downstream of RTK activation:
(1) Phosphorylated RTK recruits GRB2 (an SH2 domain adaptor protein) and SOS (a guanine nucleotide exchange factor for RAS)
(2) SOS activates RAS by promoting GDP→GTP exchange; RAS is a monomeric G protein analogous to the Gα subunit of heterotrimeric G proteins
(3) RAS-GTP activates RAF (a serine/threonine kinase)
(4) RAF phosphorylates and activates MEK
(5) MEK phosphorylates and activates ERK1/2 (the MAPKs — mitogen-activated protein kinases)
(6) ERK1/2 translocates to the nucleus and phosphorylates transcription factors, driving expression of genes governing cell cycle entry, proliferation, and survival
![]() The
clinical consequence of this pathway's central importance: RAS
mutations (most commonly KRAS G12C, G12D, G12V) are found in
approximately 30% of all human cancers.
The
clinical consequence of this pathway's central importance: RAS
mutations (most commonly KRAS G12C, G12D, G12V) are found in
approximately 30% of all human cancers.
BRAF V600E mutation (constitutively activating RAF) is found in ~50% of melanomas, and in substantial proportions of colorectal, thyroid, and other cancers. Drugs targeting this pathway at multiple levels are now in routine clinical use.1,2
 |
 |
![]() The
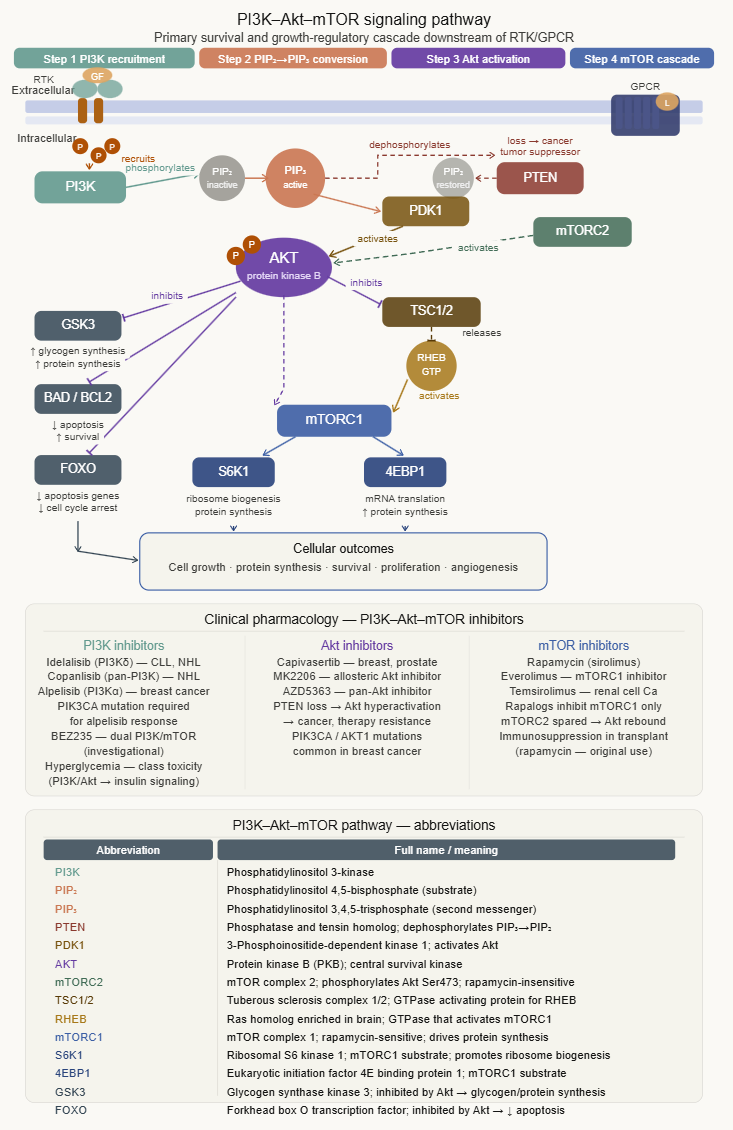
PI3K–Akt–mTOR pathway
is the primary survival and growth-regulatory cascade:
The
PI3K–Akt–mTOR pathway
is the primary survival and growth-regulatory cascade:
(1) Phosphorylated RTK recruits PI3K (phosphatidylinositol 3-kinase)
(2) PI3K phosphorylates PIP2 → PIP3 at the inner leaflet of the plasma membrane
(3) PIP₃ recruits Akt (protein kinase B) to the membrane, where it is activated by PDK1
(4) Akt phosphorylates numerous substrates controlling protein synthesis, glucose metabolism, and apoptosis inhibition
(5) mTOR (mechanistic target of rapamycin) is activated downstream, driving ribosome biogenesis and protein synthesis essential for cell growth
PTEN, the phosphatase that dephosphorylates PIP3 back to PIP2, is a tumor suppressor that negatively regulates this pathway.
Loss of PTEN function is one of the most common events in human cancer
 |
Pharmacological Targeting of RTKs: Tyrosine Kinase Inhibitors
![]() The
recognition that dysregulated receptor tyrosine kinases (RTKs)
signaling drives tumor growth led to one of the most productive
drug discovery programs in modern oncology which was the
development of tyrosine kinase inhibitors (TKIs).
The
recognition that dysregulated receptor tyrosine kinases (RTKs)
signaling drives tumor growth led to one of the most productive
drug discovery programs in modern oncology which was the
development of tyrosine kinase inhibitors (TKIs).
These drugs embody Paul Ehrlich's original magic bullet concept at the molecular level: selectively targeting the cancer-driving molecular lesion while sparing normal cells.10
TKIs fall into two structural categories
Small-molecule kinase inhibitors
These inhibitors compete with ATP for binding to the kinase domain ATP-binding pocket which prevents phosphorylation.
Because the ATP-binding pocket structure varies between kinases, selective inhibitors can be designed; however, since these inhibitors are not perfectly selective "off-target" kinase inhibition may account for both additional therapeutic effects and adverse effects.
![]() Imatinib (Gleevec) is an
excellent example of a tyrosine kinase inhibitor.
Imatinib (Gleevec) is an
excellent example of a tyrosine kinase inhibitor.
Chronic myeloid leukaemia (CML) is driven by the BCR-ABL fusion oncogene was results in a constitutively active tyrosine kinase due to the Philadelphia chromosome translocation.
 |
|
Imatinib was rationally designed to fit the ATP-binding pocket of BCR-ABL in its inactive conformation, blocking kinase activity.10
![]() Its
clinical development transformed chronic myeloid
leukemia from a disease with median survival of
3–5 years to one in which most patients achieve
complete molecular remission and near-normal
life expectancy.
Its
clinical development transformed chronic myeloid
leukemia from a disease with median survival of
3–5 years to one in which most patients achieve
complete molecular remission and near-normal
life expectancy.
Subsequent generations (dasatinib, nilotinib, ponatinib) were developed to overcome resistance mutations in BCR-ABL.10
![]() Other important
small-molecule TKIs include:
Other important
small-molecule TKIs include:
Gefitinib, Erlotinib Osimertinib
These drugs are classified as Epidermal Growth Factor Receptor (EGFR) inhibitors for EGFR-mutant non-small cell lung cancer.
Vemurafenib, Dabrafenib
These drugs are classified as BRAF V600E inhibitors for melanoma
Sunitinib, Sorafenib
Ruxolitinib, Tofacitinib
These drugs are classified as JAK inhibitors
Monoclonal antibodies targeting RTK extracellular domains represent the biologic approach to the same targets.
Trastuzumab (Herceptin) binds the extracellular domain of HER2 (ErbB2), blocking ligand-independent dimerization and signaling, and recruiting immune effector mechanisms.
Cetuximab targets the EGFR extracellular domain.
These agents differ from small-molecule Tyrosine Kinase Inhibitors in their size, half-life (weeks), and mechanisms of action beyond simple kinase blockade.
Large molecular size requires that such agents be administered parenterally.
JAK-STAT Receptors: Cytokine Signaling Without Intrinsic Kinase Activity
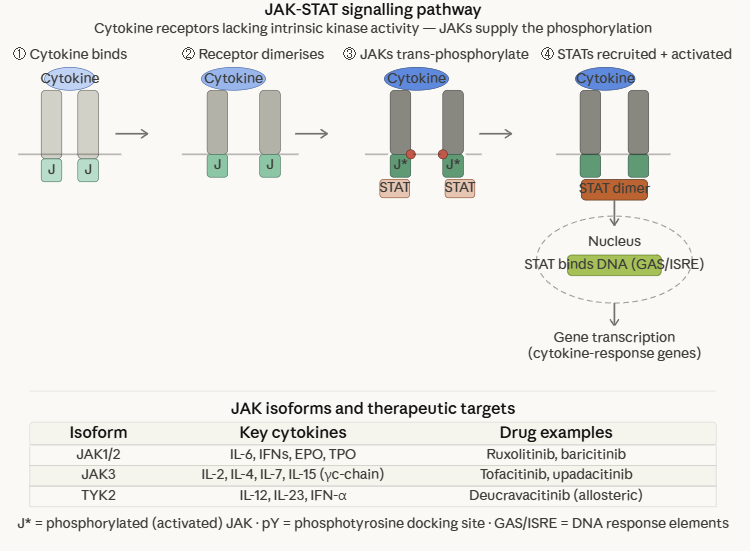
The JAK-STAT pathway is a second major class of enzyme-linked receptor signaling which differs from RTKs in an important way as these receptors themselves lack intrinsic kinase activity.
Instead, their intracellular domains are constitutively associated with Janus kinases (JAKs) which are cytoplasmic tyrosine kinases activated by receptor dimerisation.6
 |
JAK-STAT receptors are the signaling mechanism for a large family of cytokines and growth factors, including:
Interferons (IFN-α, -β, -γ)
Interleukins (IL-2, -4, -6, -7, -12, -13, -15, -23, and many others)
Erythropoietin
Thrombopoietin
Growth hormone, and
Prolactin
Activation mechanism
(1) Cytokine binding induces receptor dimerization (or oligomerization), bringing two receptor-associated JAK molecules into proximity
(2) Trans-phosphorylation activates the JAK kinases
(3) Activated JAKs phosphorylate specific tyrosine residues on the receptor intracellular domain, creating docking sites for STAT proteins (Signal Transducers and Activators of Transcription)
(4) STATs bind these phosphotyrosine docking sites via their SH2 domains, where they are phosphorylated by JAKs
(5) Phosphorylated STATs dimerize and translocate to the nucleus, where they directly bind DNA and activate transcription of cytokine-response genes6,7
The four JAK isoforms (JAK1, JAK2, JAK3, TYK2) pair with different cytokine receptor families, providing the selectivity basis for therapeutic JAK inhibition:
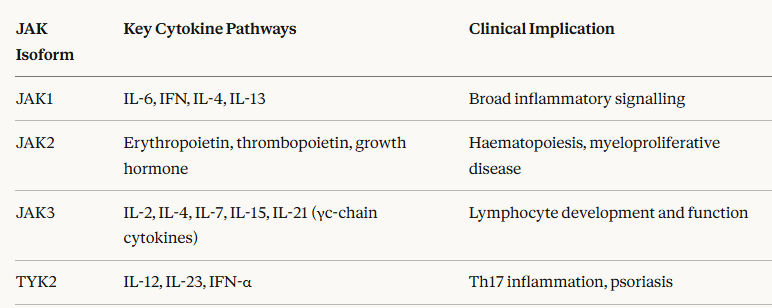 |
JAK inhibitors (Jakinibs) are approved for multiple inflammatory and hematological conditions:7
Ruxolitinib (JAK1/2)
Myelofibrosis, polycythaemia vera, acute and chronic GVHD
Tofacitinib (JAK1/3)
Rheumatoid arthritis, psoriatic arthritis, ulcerative colitis
Baricitinib (JAK1/2)
Rheumatoid arthritis, atopic dermatitis, COVID-19 (hospitalised patients)
Upadacitinib (JAK1-selective)
RA, psoriatic arthritis, atopic dermatitis, Crohn's disease
Deucravacitinib (TYK2)
Psoriasis treatment based on novel allosteric mechanism targeting the regulatory domain rather than the ATP-binding site.
The class effect adverse profile of JAK inhibitors reflects their mechanism:
Immunosuppression resulting in increased susceptibility to bacterial, viral, and opportunistic infections, including reactivation of latent herpes zoster
Hematological effects resulting in anemia, thrombocytopenia from JAK2 inhibition
Cardiovascular and thromboembolic risk (possible) possibly leading to regulatory warnings requiring consideration of patient risk factors before prescribing.7
Nuclear Receptors: Intracellular Transcription Factor Receptors
Nuclear receptors (NRs) constitute a superfamily of intracellular ligand-activated transcription factors that directly regulate gene expression.
Their ligands are lipophilic small molecules capable of crossing the cell membrane by passive diffusion and include steroid hormones (glucocorticoids, mineralocorticoids, sex steroids), thyroid hormones, vitamin D₃, retinoids (vitamin A derivatives), and bile acids.8
The human genome encodes 48 nuclear receptors.
They are classified into two subfamilies:
Type I nuclear receptors (steroid hormone receptors)
These receptors are found in the cytoplasm in an inactive complex with heat shock proteins (HSP90, HSP70) in the absence of ligand.
Ligand binding causes dissociation of HSP chaperones, receptor homodimerization, and nuclear translocation.
Type I include: glucocorticoid receptor (GR), mineralocorticoid receptor (MR), androgen receptor (AR), estrogen receptor (ER), and progesterone receptor (PR).
Type II nuclear receptors (thyroid hormone, retinoid, vitamin D receptors)
These receptors are found constitutively in the nucleus, typically as heterodimers with the retinoid X receptor (RXR).
In the absence of ligand they associate with co-repressor complexes (NCOR, SMRT) that maintain chromatin in a compacted, transcriptionally repressed state.
Type II receptors include thyroid hormone receptors (TRα, TRβ), retinoic acid receptors (RARα, -β, -γ), vitamin D receptor (VDR), and peroxisome proliferator-activated receptors (PPARα, -γ, -δ).8,9
Mechanism of Nuclear Receptor Action
The molecular mechanism of nuclear receptor action. from ligand binding through to altered gene transcription, is one of the most studied and best understood signaling pathways in all of biology.
This conclusion is likely justified because this nuclear receptor activity represents the mechanism of the glucocorticoids, the most widely prescribed anti-inflammatory drugs in clinical medicine.9
Core mechanism (using Glucocorticoid Receptor as the example)
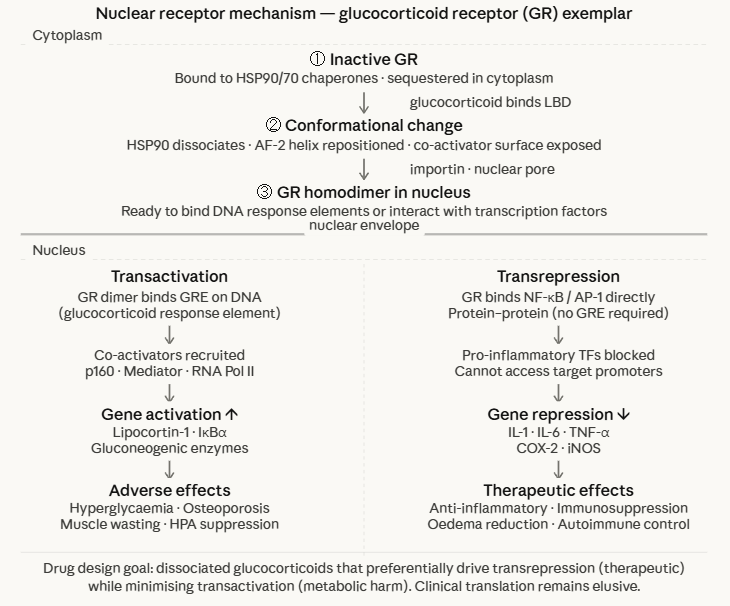 |
Ligand entry:
A glucocorticoid such as cortisol, dexamethasone, prednisolone diffuses across the cell membrane and binds the ligand-binding domain of the cytoplasmic glucocorticoid receptor.
Conformational change
Ligand binding induces a major conformational change in the ligand-binding domain, particularly repositioning of helix 12 (the activation function-2 helix, AF-2), that simultaneously dissociates HSP90/70 chaperones and creates a hydrophobic surface for co-activator recruitment.
Nuclear translocation
The ligand-bound glucocorticoid receptor homodimerizes and is imported into the nucleus through nuclear pores via importin proteins
DNA binding:
The glucocorticoid receptor dimer binds specific DNA sequences called glucocorticoid response elements (GREs), palindromic 15 bp sequences located in the promoter and enhancer regions of target genes.
[Strahle U Klock G Schutz YERVOY one DNA sequence of 15 base pairs is sufficient to mediate both glucocorticoid and progesterone induction of gene expression. Proc Nat Acad Sci U S A 1987 November;84(22): 7871-7075. Click for Abstract]
The glucocorticoid receptor DNA-binding domain (DBD) contains two zinc finger motifs that grip the GRE major groove
Transcriptional regulation: Ligand-bound GR can either activate or repress gene transcription, through distinct mechanisms:
Transactivation (GRE-dependent gene activation)
Ligand-bound GR at GREs recruits co-activator complexes, including p160 co-activators (SRC-1, TIF2, AIB1) and the mediator complex, that remodel chromatin and recruit RNA polymerase II, activating transcription.
Genes induced by glucocorticoids include those encoding:
lipocortin-1 (annexin-1, which inhibits phospholipase A₂)
IκBα (which inhibits NF-κB)
Gluconeogenic enzymes
Transactivation is responsible for many of the metabolic adverse effects of glucocorticoids (hyperglycaemia, muscle wasting, osteoporosis).9
Transrepression (GRE-independent gene repression)
Ligand-bound GR physically interacts with and inhibits pro-inflammatory transcription factors, particularly NF-κB and AP-1 (activator protein 1, preventing them from activating their target gene promoters.
[Bhosale P Kim H Abusaliya A Vetrivel P Ha S Park M Lee H Kim G Structural and Functional Properties of Activator Protein-1 in Cancer and Inflammation. Evid Based Complement Alternat Med 2022 May 26;2022: Click for Article]
This protein-protein interaction does not require GR to bind DNA directly.
Transrepression is the mechanism responsible for the primary anti-inflammatory and immunosuppressive effects of glucocorticoids and suppression of cytokine genes (IL-1, IL-6, TNF-α, COX-2).9
Clinical Pharmacology of Nuclear Receptor Ligands
Slow onset
Effects require hours to days because new protein synthesis is the effector mechanism and mRNA must be transcribed, exported, translated, and the resulting proteins must accumulate to functionally significant levels.
This sequence of events explains why glucocorticoids do not provide immediate relief in acute anaphylaxis and why associated anti-inflammatory effects in conditions like asthma or rheumatoid arthritis develop over days.
Prolonged duration
Because newly synthesized proteins persist after the drug is eliminated, the duration of action of nuclear receptor drugs considerably exceeds their pharmacokinetic half-life.
A single dose of dexamethasone has pharmacological effects lasting 24–36 hours despite a plasma half-life of only 3–5 hours.
Broad genomic effects
Nuclear receptors regulate hundreds to thousands of genes.
Glucocorticoids regulate genes involved in immunity, metabolism, bone turnover, skin integrity, mood, and adrenal axis function simultaneously.
![]() This breadth of genomic effect is the mechanistic basis
for the extensive adverse effect profile of systemic
glucocorticoids.
This breadth of genomic effect is the mechanistic basis
for the extensive adverse effect profile of systemic
glucocorticoids.
Genomic cannot be fully separated from their therapeutic effects by dose adjustment alone.1,2
HPA axis suppression and adrenal insufficiency
Exogenous glucocorticoids suppress hypothalamic CRH and pituitary ACTH secretion through negative feedback at nuclear GRs in both tissues.
With prolonged use, the adrenal cortex atrophies from lack of ACTH stimulation.
Abrupt discontinuation of long-term glucocorticoid therapy can precipitate adrenal crisis, an inability to mount a cortisol stress response.
Adrenal crisis representas a pharmacodynamic consequence of nuclear receptor-mediated HPA suppression, not a pharmacokinetic phenomenon.
Gradual tapering allows recovery of HPA axis function.
|
|
|
|
|
|
|